
In the evolving landscape of crude oil processing, refiners are increasingly turning to opportunity crudes to boost profitability. The lifting of the US export ban in 2015 opened up a broader range of feedstocks for Asian refiners, particularly US-origin crudes like WTI. While these crudes are often traded at a discount, making them economically attractive to process, the presence of contaminants such as Fe and Ca pose additional challenges for refiners. In this article, we explain the challenges due to high levels of iron (Fe) and calcium (Ca), detailing the contamination mechanism, diagnostic tools and solutions to mitigate it.
Mechanism of Iron (Fe) Poisoning on Catalyst
Contaminant Fe in FCC operation can be broadly classified into two main categories: “Molecular/Colloidal” Fe and “Particulate/Tramp” or Inorganic Fe.
Molecular/Colloidal Fe, otherwise also known as ‘Organic Fe,’ is typically bound to the hydrocarbon molecule and deposits on the outer periphery of the catalyst, exhibiting limited intra-particle mobility. Ketjen’s fundamental work, supported by Scanning Electron Microscopy (SEM), has shown that organic Fe concentrates within the outer 2-5 µm layer of the catalyst particle, forming a distinct ring (Figure 1, left). This accumulation forms a ‘skin’ that obstructs pore openings, hindering the diffusion of large hydrocarbon molecules to the catalyst’s active sites and decreasing accessibility, thereby reducing overall FCC conversion and negatively impacting slurry yields. At the same time, it limits the egress of molecules out of the catalyst interior causing unselective catalytic reactions leading to dry gas and coke. Also, hydrogen transfer is enhanced which thereby converts valuable light olefins into paraffins.
Calcium (Ca) has a similar deposition profile to iron, forming rings of high Ca concentration on the external particle surface (Figure 1, right). Additionally, calcium, being an alkaline earth element, can have some basicity, which can neutralize catalyst active sites, lowering catalyst activity depending on the concentration level. Furthermore, in the severe hydrothermal conditions of the regenerator, iron deposited on the catalyst can interact with calcium, sodium, and silica, resulting in the formation of low melting eutectics. Once these phases form, the catalyst surface vitrifies, resulting in the formation of nodules and causing permanent damage. Surface pores close, preventing feed molecules from reaching interior cracking sites, which results in reduced catalyst activity and conversion, and an increase in bottoms yield. Additionally, ABD decreases with nodule formation and depending on the intensity of nodule formation, negatively affects the catalyst fluidization properties. Severe deterioration in ABD could pose a risk of losing circulation and lead to an unplanned shutdown.
Particulate/Tramp Fe is typically inorganic in nature, mainly resulting from the corrosion of refinery hardware. Additionally, soil/clay particulates found in crude oil from enhanced oil recovery techniques can also manifest in FCC operations. Typically, this category of Fe is broadly considered to have a limited impact on FCC catalyst performance. However, in some cases, and depending on the unit design, particulate Fe can contribute to the formation of nodules on the catalyst, impact the fluidization and catalyst circulation in the unit, and have undesirable effects on controlling CRC in partial burn units and SOx emissions.
Given the variable types of Fe and impacts on the FCCU operation, Ketjen has developed a set of diagnostic tools for refiners to understand the nature and impact of Fe poisoning.


Diagnosing Iron (Fe) Contamination on Catalysts
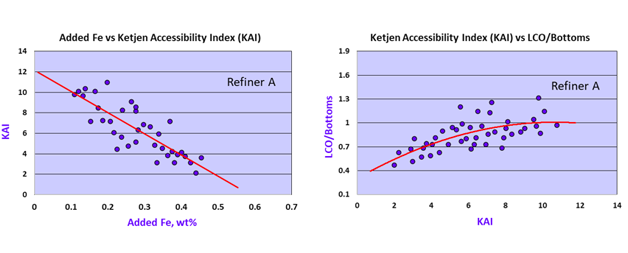
Over the years, the technical community has explored various methods to quantify the impact of Fe and Ca on FCC catalysts, which are known to deteriorate pore structure and restrict hydrocarbon access to the active sites. To overcome the limitations of traditional techniques, Ketjen developed the proprietary Ketjen Accessibility Index® (KAI), which accurately quantifies the dynamic diffusional capacity of fresh FCC and equilibrium catalysts. This proprietary test measures and quantifies the dynamic diffusivity of a large hydrocarbon molecule into the catalyst active sites. A commercial example from Refiner A illustrates how Fe contamination directly reduces accessibility in catalysts (Figure 3, left). Conversely, high accessibility catalysts reduce slurry yield (LCO/Bottoms ratio) while facing challenges from Fe contamination (Figure 3, right). Furthermore, in addition to the KAI analysis, Ketjen has developed a series of proprietary diagnostic techniques to understand and troubleshoot Fe and Ca contamination-related FCC issues.
Solution to Mitigate Fe Contamination
Over the past few decades, Ketjen has provided high accessibility catalyst technologies known for their performance in mitigating the harmful impact of Fe and Ca by elevating the accessibility of Ecat for given levels of contaminant Ca and Fe, thereby minimizing yield deterioration and margin loss. All our proven families of catalysts, such as DENALI®, AFX™, ACTION®, and UPGRADER™, are manufactured using the high KAI platform and have been benchmarked to have best-in-class Fe and Ca tolerance.
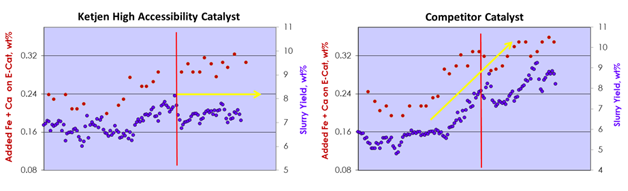
A commercial Resid FCC unit demonstrates how Ketjen’s high accessibility catalyst effectively mitigates the impacts of Fe contamination. In the unit trial, Ketjen’s high accessibility catalyst exhibited no increase in slurry yield when added Fe and Ca on E-Cat was increased by +1000 ppm. Conversely, a competitive catalyst showed a significant increase in slurry yield of +2.5 wt% for a similar level of increase in added Fe and Ca on E-Cat.
With increasing levels of contaminant Fe and Ca in E-Cat, driven by elevated processing of Fe/Ca-bearing crudes such as WTI, Ketjen is preparing to introduce a breakthrough catalyst that delivers step-out Fe and Ca tolerance on top of our existing high KAI technology. Stay tuned for our next post, where Ketjen experts will explain how Ketjen’s breakthrough technologies can help refiners take advantage of the opportunity feedstocks to enhance margins while managing Fe and Ca.